They Retracted the 2025 Paper That Proved Fenbendazole Cures Cancer
Here’s Why That Tells You Everything & ⚡️MASSIVE WEEKEND FLASH SALE CONTINUES ⚡️
This Substack’s readers have known for many years now that Fenbendazole cures cancer…
…and now we have further evidence that the Medical-Industrial Complex, their BigPharma partners-in-crime and their captured researchers are desperate to keep the most effective, safest and least expensive cancer cure away from everyone; to wit:
by William F. Supple, Jr., Ph.D.
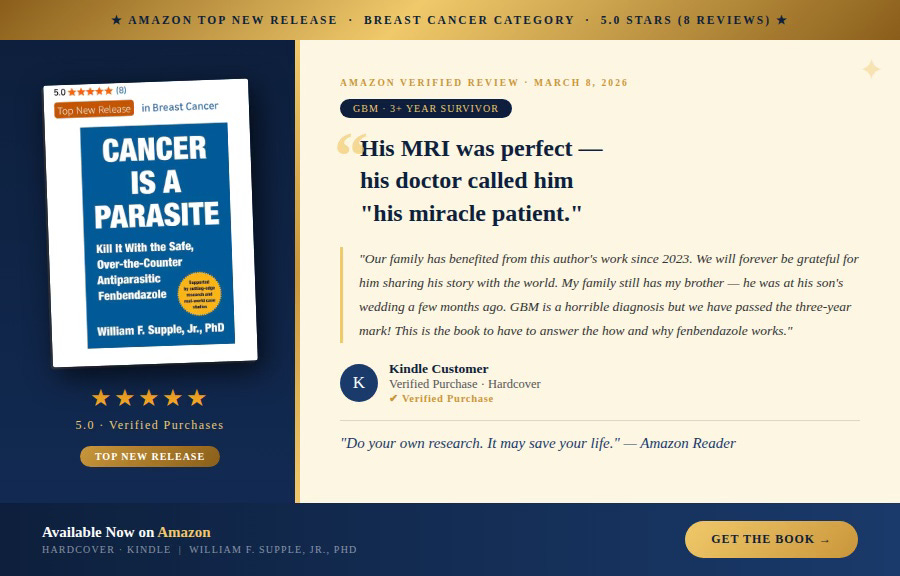
Fenbendazole Can Cure Cancer presents Case Reports of people who have treated their own cancers along with other articles to help understand how fenbendazole works to treat cancer. Previous articles covering other cancers are in the Archives link.
Big Cancer tipped its hand — and showed its true colors.
A medical journal just retracted one of the most significant papers ever published on fenbendazole as a cancer treatment. Three patients. Three different solid tumor metastatic cancers. All three in complete or near-complete remission. No adverse effects. Sustained over years.
That paper is now gone from the literature (it is available for download as a PDF but its content are not indexed or searchable on PubMed). And the reason given for pulling it is one of the most cynical acts of institutional overreach on record.
I want to tell you exactly what happened — and why it matters far more than the retraction itself. I took this retraction personally, not because I was an author, but because I supplied all the case report data that comprised the paper. One case is my mother-in-law, one has become a good friend and another is a stellar individual always ready to help others. They submitted their information faithfully to help others and that effort was stolen from them.
The Paper
Published in Case Reports in Oncology in May 2025, Makis et al. documented three patients with Stage IV cancers — breast, prostate, and melanoma — who achieved dramatic and durable remissions after incorporating fenbendazole into their treatment regimens. Two achieved complete remission. One near-complete remission. All three tolerated fenbendazole without any reported adverse effects. All three remained in remission through the entire follow-up period, ranging from 11 months to nearly three years (Makis et al., 2025).
The lead author was Dr. William Makis, an oncologist who has treated cancer patients with fenbendazole and other repurposed antiparasitic drugs. His paper passed peer review. It was accepted and published. It entered the medical literature.
And then it was retracted.
The “Reason” — and Why It Is Fabricated
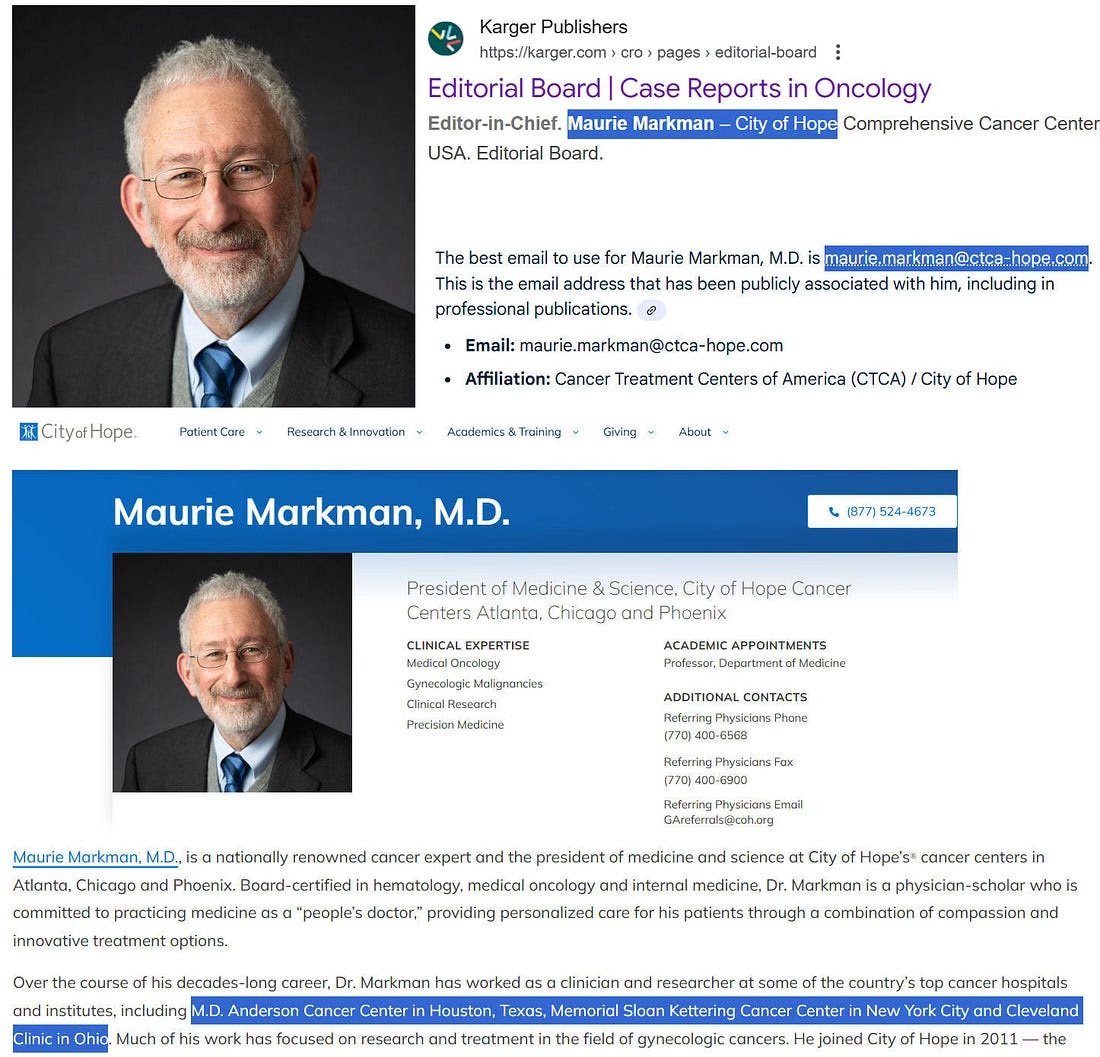
The retraction was ordered by the journal’s Editor, Dr. Maurie Markman, on the grounds that Dr. Makis had an undeclared financial interest — specifically, that at the time of submission he offered services related to the topic of the study, i.e., he treats cancer patients with fenbendazole. The retraction statement noted that “the author’s response when contacted were found to be unsatisfactory” and concluded that “these concerns would have affected the interpretation of the work” (Case Reports in Oncology, 2026).
Read that again and let it sink in.
fenbendazole is an off-patent, over-the-counter veterinary antiparasitic. It costs pennies per dose. No pharmaceutical company owns exclusive rights to it. No one profits handsomely from its use. As reviewed by Nguyen et al. (2024), fenbendazole’s patent expired in the early 1990s, making it available as a generic drug accessible through animal supply stores and online platforms. The “financial interest” claimed against Dr. Makis — a physician helping patients with a drug that generates essentially no revenue — is not just weak. It is nonsensical.
Now let’s talk about who Dr. Markman actually is and who actually has a financial conflict of interest here.
The Real Conflict of Interest
Dr. Maurie Markman is the President of Medicine and Science at City of Hope Cancer Center. City of Hope is a multi-billion-dollar cancer treatment enterprise that generates its revenue from — you guessed it — cancer treatment. Expensive cancer treatment. Patented cancer treatment. Chemotherapy, immunotherapy, targeted biologics: treatments that cost hundreds of thousands of dollars per patient per year, that are frequently toxic, that often extend life by only weeks to months, and that leave behind a devastating legacy of cancer stem cells that drive relapse and metastasis.
Dr. Markman, in his role as Editor in Chief of Case Reports in Oncology, approved this paper for publication. It passed peer review. He read it. He accepted it. The science was sound. And then something changed.
What changed? The paper gained attention, lots of attention. It documented exactly what thousands of cancer patients around the world using fenbendazole already know: it works. It works remarkably well. And it cannot be monetized by Big Cancer institutions like City of Hope.
The financial conflict of interest here is not Dr. Makis’s. It is Dr. Markman’s. The institutions that retracted a paper proving that a cheap, off-patent drug cures cancer are the same institutions that profit enormously from ensuring that patients remain dependent on expensive, patent-protected treatments.
The irony of the name “City of Hope” is almost too rich to bear.
The Hypocrisy Is Breathtaking
Let us apply Dr. Markman’s financial conflict-of-interest standard consistently. If we were to scrutinize every paper published in oncology journals by researchers employed by pharmaceutical companies, by cancer centers compensated for clinical trial enrollment, by investigators receiving speaking fees and consulting payments from drug manufacturers — no paper would survive. The entire edifice of modern oncology research is saturated with financial conflicts of interest that make Dr. Makis’s alleged infraction look like a parking ticket.
Here is what is true: because Dr. Makis uses a cheap, off-patent drug with no commercial value, his paper is the only paper that would genuinely pass a rigorous financial conflict-of-interest standard. The retraction inverts reality. It punishes this one researcher in the field who has no financial incentive to lie.
If there is an Editor that would be interested in publishing this impeccably-researched, peer-reviewed ground-breaking article please reach out to either myself or Pierrick Martinez.
The Cancer Stem Cell Problem — and Why This Matters
There is a scientific dimension to this story that receives almost no attention in mainstream oncology, but which I detail at length in my book Cancer is a Parasite.
Conventional cancer treatments — the chemotherapy and radiation administered at places like Cancer Centers — are extraordinarily effective at killing the bulk of a tumor. What they are not effective at killing, and what they may actually stimulate and expand, are cancer stem cells: the treatment-resistant, highly plastic cells that survive conventional therapy and seed recurrence and metastasis (Tu et al., 2024).
Fenbendazole kills cancer stem cells. This has been documented in multiple peer-reviewed studies. In a 2025 publication in Molecules, Lei et al. demonstrated that fenbendazole dose-dependently inhibited proliferation and triggered apoptosis in both conventional cervical cancer cells and cervical cancer stem cells — including the CD133+CD44+ stem cell population that drives therapy resistance and metastasis (Lei et al., 2025). The mechanism involved enforcing G2/M blockade through modulation of cyclin B1, cdc25C, Wee1, and CDK1 — a multi-target disruption of the cell cycle machinery in the very cells that survive standard treatment.
As also documented in the Makis et al. case series itself, benzimidazoles including fenbendazole have been shown to affect cancer stem cells (Makis et al., 2025). The broader anticancer mechanisms of fenbendazole — p53 activation, GLUT1 inhibition, hexokinase suppression, glucose uptake reduction, microtubule disruption, and proteasomal impairment — are comprehensively reviewed in Nguyen et al. (2024).
In other words: the treatments that institutions like Cancer Centers administer create the very cells that kill patients, and fenbendazole eliminates those cells. Dr. Markman’s institution is not a passive bystander in this story. It is an active participant in a treatment model that fenbendazole threatens to render obsolete.
This Playbook Is Not New
What Dr. Markman has done is not novel. It follows a well-documented playbook of suppression that I trace back at least to 1976 in Cancer is a Parasite. The tools are familiar: delegitimization, deception, procedural pretext, journal pressure, manufactured conflicts of interest. The goal is always the same — to prevent cheap, unpatentable treatments from gaining clinical traction.
But this time, I believe they have made a grave strategic error.
They tipped their hand. They retracted the most definitive published case series ever demonstrating that fenbendazole produced complete remission across three types of metastatic solid tumors. They did this in full view of a public that is increasingly literate about antiparasitic drugs, increasingly skeptical of Big Pharma, and increasingly motivated to seek alternatives. And, perhaps most importantly, in a political environment that may not let them get away with it any longer.
The 590 documented case reports of fenbendazole, ivermectin, and mebendazole in cancer— compiled by OneDayMD.com as of March 2026 — do not disappear because a journal editor under pressure pulled one paper. The science does not disappear. The patients who are alive today because of fenbendazole do not disappear.
And neither does the clinical trial data. At the 2025 ASCO Annual Meeting, Bitar et al. reported Phase I/II results (NCT05318469) combining ivermectin with the checkpoint inhibitor balstilimab in metastatic triple-negative breast cancer: a 37.5% clinical benefit rate in heavily pretreated patients, with no dose-limiting toxicities (Bitar et al., 2025). Antiparasitic drugs are entering the clinical trial pipeline whether Big Cancer likes it or not. Notably, this trial was previously run out of City of Hope before transferring to Cedars-Sinai — an additional irony worth noting in a future post.
The Population-Level Evidence that Antiparasitics Appear to Also Prevent Cancer That They Also Cannot Explain
According to publicly available WHO data, the 123 nations that conduct mass drug administration (MDA) programs — administering antiparasitic drugs including benzimidazoles to their populations at scale — have approximately half the cancer incidence of wealthy nations that do not have such programs. Billions of doses of antiparasitic drugs are administered annually under these government-run programs. The safety record is unassailable (Supple, 2026).
This is not a fringe claim. It is WHO-documented population-level data. And it is data that the current cancer research apparatus has no interest in exploring, because exploring it leads directly to fenbendazole.
What Must Happen Now
The safety question for antiparasitics in cancer has already been answered — by billions of administrations of doses each year across 123 countries. This is not a new drug. The concept of using antiparasitic agents against cancer is not new either: the Soviets apparently understood it in the 1930s, other researchers rediscovered it in 1976, and the world is rediscovering it again today.
What is needed now is a committed, government-directed, independent, stooge-free research program into antiparasitics as cancer treatments — a moon-shot type effort to eradicate deaths from most forms of cancer within the next few years. We already have the drugs. We know why they work: Cancer is a Parasite. These drugs have been inadvertently safety tested for many years during their primary use as antiparasitics. We just need to go the last mile: optimize the administration, bioavailability, dosing, manufacture and distribution of these drugs. It would be useful to know if there are any circumstances where fenbendazole is unlikely to be effective. Cancer patients given up for dead after traditional treatments failed are saving their own lives now with fenbendazole and other repurposed drugs. Let’s help them out, and others yet to be affected, with fairly researched answers and useful solutions.
This program should be housed within the Department of Health and Human Services and must be conducted without the corrupting influence of the pharmaceutical industry and the institutional cancer establishment. The standard cancer research apparatus cannot be trusted to conduct this work. The retraction of the Case Reports in Oncology paper proves it. They will kill the baby in the cradle, as they have done before. They showed us exactly who they are.
Secretary Kennedy and the Department of Health and Human Services now have an opportunity — and an obligation — to act. The MAHA mandate is precisely this: to pursue health solutions that actually serve the American people, not the financial interests of entrenched institutions.
The evidence is in. The safety is established. Three cancer patients are alive and thriving today who might not be otherwise. And the editor who tried to erase them from the medical record has a multi-billion-dollar conflict of interest that dwarfs anything he accused Dr. Makis of.
It is time to act.
Indeed, the time to act is now, and the following treatment approach represents not only the ‘holy grail’ (turbo) cancer cure in plain sight, but may also treat “vaccine” shedding exposure, Alzheimer’s, mood disorders, Parkinson’s, Lyme Disease, myocarditis, Hashimoto’s Disease, leukemia, Lupus, skin conditions, and various other “incurable” ailments, as well as the common cold and seasonal flu:
The Ultimate Disease Cure & Prophylaxis Protocol
Tocotrienol and Tocopherol forms (all 8) of Vitamin E (400-800mg per day, 7 days a week). A product called Gamma E by Life Extension or Perfect E are both great.
Bio-Available Curcumin (600mg per day, 2 pills per day 7 days a week). A product called Theracurmin HP by Integrative Therapeutics is bioavailable.
Vitamin D (62.5 mcg [2500 IU] seven days a week).
CBD oil (1-2 droppers full [equal to 167 to 334 mg per day] under the tongue, 7 days a week) CBD-X: The most potent full spectrum organic CBD oil, with 5,000 milligrams of activated cannabinoids and hemp compounds CBD, CBN & CBG per serving.
Fenbendazole (300mg, 7 days a week) or in the case of severe turbo cancers up to 1 gram — for prophylaxis one 150mg tablet once or twice per week
Ivermectin (24mg, 7 days a week) or in the case of severe turbo cancers up to 1mg/kg/day — for prophylaxis one 12mg tablet once or twice per week
Hydroxychloroquine (10mg/kg/day 7 days a week) - for prophylaxis one 200mg tablet once or twice per week
Doxycycline (100mg, 7 days a week for 30-60 days)
VIR-X immune support which also greatly increases the bioavailability of both Fenbendazole and Hydroxychloroquine (2 capsules per day) — for prophylaxis 2 capsules per day
Removing sugars and carbohydrates (cancer food) from your diet and replacing table sugar with a zero glycemic index, zero calorie, keto friendly rare sugar like FLAV-X
Do NOT comply.
⚡️ THE MASSIVE WEEKEND FLASH SALE starts now, so please use code FLASH25 to receive 25% off on the newest product Hydroxychloroquine, as well as ALL of the amazing lifesaving products that you have been purchasing for many years now like the Nobel Prize winning miracle compound Ivermectin, the no less miraculous Fenbendazole, Doxycycline, the full spectrum organic CBD oil containing 5,000 milligrams of activated cannabinoids and hemp compounds CBD, CBN & CBG, the powerful immune support nutraceutical and spike support formula VIR-X, and the sugar craving reducing, blood sugar balancing and even anti-cancer allulose sugar substitute FLAV-X! ⚡️
THE MASSIVE 25% OFF LIFESAVING FLASH SALE ends Sunday, March 22nd (midnight eastern time), 2026.
Upon adding products to your cart, please go to the cart icon at the top right corner of your browser page and click it, then choose the VIEW CART option whereby you will be redirected to a page where you can enter the code FLASH25 in the Use Coupon Code field.
Only the RESOLVX HEALTH website offers all of the authentic products that this Substack promotes.
Please contact the company directly with any product questions: info@resolvx.health







What else would you expect when the cancer business in the USA is worth $200 billion and finding cheap alternatives to solving the problem is going to impact the income of the oncologist and all the drug manufacturers that sell high price chemo drugs at a rate seven times higher than Europe with the same health outcomes.
Your protocol has a lot of supplements and other medicinals. How do you know that every one of them is necessary? If FenBen is the one that does the trick, why not take just fenben? If the others are necessary, then the cancer is not defeated by Fenben, it's defeated by all of the above in combination.
Regarding the paper that was retracted, what protocol did Dr. Makis use to treat the 3 patients? Or to put it more directly, were the patients taking the protocol that you lay out here on your substack, or some varation of that? Did they also undergo chemo or radiation or both? Why did Dr. Makis conclude that it was Fenben that cured the cancer and not any of the other things the patients were taking and doing?