Dr. Paul Marik's New Substack: Cancer is a Metabolic Disease
‼️ AN ALTERNATE THEORY OF CANCER: CANCER IS A METABOLIC DISEASE & THE 25% OFF WEEKEND FLASH SALE ENDS TONIGHT ‼️
Dr. Paul Marik is a bonafide hero of the medical freedom movement, and just the other day he started his very own Substack publication called, Cancer & Metabolic Healing, which everyone should consider subscribing to.
For many years now this Substack has been proudly featuring the good doctor across a variety of health related subjects; for example:
Etc. & etc. & etc.
And now Dr. Marik has just published an important article which will be accompanied by a treatment protocol that longtime readers of this Substack are well aware of.
by Cancer & Metabolic Healing/Dr. Paul Marik
A patient who felt healthy only weeks before now sits in a quiet examination room, waiting to hear the results of a biopsy. The words that follow are simple but devastating: “You have cancer.” In that moment the world seems to contract. Questions flood the mind — How did this happen? Why me? What does this mean for my future?
For more than half a century, medicine has answered these questions with a single dominant explanation: cancer is a disease of damaged genes. Mutations accumulate in the DNA of a cell, the normal controls that regulate growth fail, and the cell begins dividing uncontrollably. This genetic theory of cancer has shaped modern oncology and driven vast investments in research aimed at identifying the mutations responsible for each tumor.
For decades, the dominant explanation has been that cancer is primarily a genetic disease — a consequence of accumulated mutations in the DNA of a single cell. This idea has guided billions of dollars of research and shaped much of modern cancer therapy. Yet despite enormous scientific advances, the overall burden of cancer continues to rise, and many treatments remain toxic, expensive, and often only partially effective.
This reality forces us to ask an uncomfortable but necessary question: Have we been looking at cancer through the wrong lens?
In recent years, a growing body of scientific evidence has begun to challenge the traditional mutation-centric view of cancer. While genetic abnormalities are clearly present in tumors, they may not be the primary driver of the disease. Instead, many investigators are returning to a much older idea — first proposed nearly a century ago — that cancer may fundamentally be a disease of disordered cellular metabolism.
At the center of this idea lies the mitochondrion, the tiny energy-producing structure within every cell. When mitochondria function normally, cells generate energy efficiently and maintain tight control over growth and division. But when mitochondrial metabolism becomes impaired, cells may revert to more primitive metabolic programs that favor uncontrolled proliferation — the biological hallmark of cancer. This shift toward glucose-driven fermentation, even in the presence of oxygen, was first described by Otto Warburg in the 1920s and is now recognized as one of the most consistent features of cancer cells.
Viewing cancer through a metabolic lens changes the conversation in profound ways. It helps explain why such diverse environmental factors — from toxins and radiation to chronic inflammation and viral infections — can all lead to malignancy. It also suggests that many of the genetic abnormalities observed in tumors may actually be downstream consequences of metabolic dysfunction rather than the original cause of the disease.
Perhaps most importantly, this perspective opens the door to new approaches for prevention and treatment. If cancer cells depend on abnormal metabolic pathways for survival, then targeting those pathways may represent a powerful therapeutic strategy.
This chapter explores the scientific foundation of the metabolic theory of cancer. We will examine the evidence supporting mitochondrial dysfunction as a central driver of tumor development, the metabolic pathways that sustain malignant growth, and the implications of this paradigm for the prevention and treatment of cancer.
Understanding cancer begins with understanding how cells generate and use energy. And as we will see, when cellular energy metabolism goes wrong, the consequences can be profound.
Understanding cancer begins with understanding how cells produce and use energy. When this fundamental process goes wrong, the consequences can be profound.
For that reason, I encourage you to read this chapter carefully and reflect on the ideas presented here. Many of the posts that follow build directly on these concepts, making this discussion a critical foundation for everything that comes next.
A basic tenet in medicine is that to treat a disease, one needs to understand the disease. Cancers are, simply, a disease of uncontrolled cell growth and division, wherein the various natural processes for containing them have partially or completely failed.
The conventional theory is that cancer is caused by genetic mutations and/or genomic instability, which drives a population of cells with the following six “classic” biological properties: (1)
Sustaining proliferative signaling
Evading growth suppressors
Resisting cell death (apoptosis)
Enabling replicative immortality
Inducing angiogenesis
Activating invasion and metastasis
Hanahan and Weinberg, who elucidated these “hallmarks of cancer,” excluded the most important and universal finding in all cancer cells, (2) namely the metabolic reprogramming of cancer cells, with aerobic glycolysis — the so-called “Warburg effect” that we will explore below. (3, 4)
Conventional thinking suggests cancer arises from a single cell due to specific mutations in that cell, which are then characteristic of the patient’s “cancer genome.” The loss of genomic “caretakers” or “guardians,” involved in sensing and repairing DNA damage, has been proposed to explain the increased mutability of tumor cells. The loss of these caretaker systems allows genomic instability, thus enabling pre-malignant cells to reach the six essential hallmarks of cancer.
The Cancer Genome Atlas Program (TCGA), modeled after the human genome project, was an attempt to determine the characteristic mutations of common cancers.(5) The TCGA assessed mutational signatures using 84,729,690 somatic mutations from 4,645 whole-genome and 19,184 exome sequences that encompassed most types of cancer. (6, 7) The findings of this massive project raises serious doubts concerning the mutation theory of cancer.
The TCGA identified 49 single-base-substitutions, 11 double-base-substitutions, 4 clustered-base-substitutions, and 17 small insertion-and-deletion signatures. However, no specific mutation was characteristic of any particular cancer (except CML and the Philadelphia chromosome). In many tumors no mutation was found, and there was marked heterogeneity of mutations between tumors of the same cell type (intertumoral heterogeneity) and within the same tumor (intratumoral heterogeneity). (8) In pediatric tumors such as medulloblastoma, the number of driver genes was low (zero to two). In common adult tumors, such as pancreatic, colorectal, breast, and brain cancers, the number of mutated driver genes was frequently between three to six, but several tumors had only one or two driver mutations. The notion that cancer is caused solely by mutations to key genes is becoming harder to maintain. (8) The inconsistencies are too numerous and pronounced.
AN ALTERNATE THEORY: CANCER IS A METABOLIC DISEASE
Travis Christofferson, in his book entitled “Tripping over the Truth”, articulated the following:
“No researcher can point to any single mutation or combination of mutations and say with confidence that it is alone the cause of cancer. Nor can researchers point to a series of cellular systems rendered dysfunctional by mutations and make the same claims with confidence.” (8)
In a 2009 op-ed for The New York Times, James Watson, a Nobel Prize winner known as the “father of DNA,” suggested that “we may have to turn our main research focus away from decoding the genetic instructions behind cancer and toward understanding the chemical reactions within cancer cells.” (9)
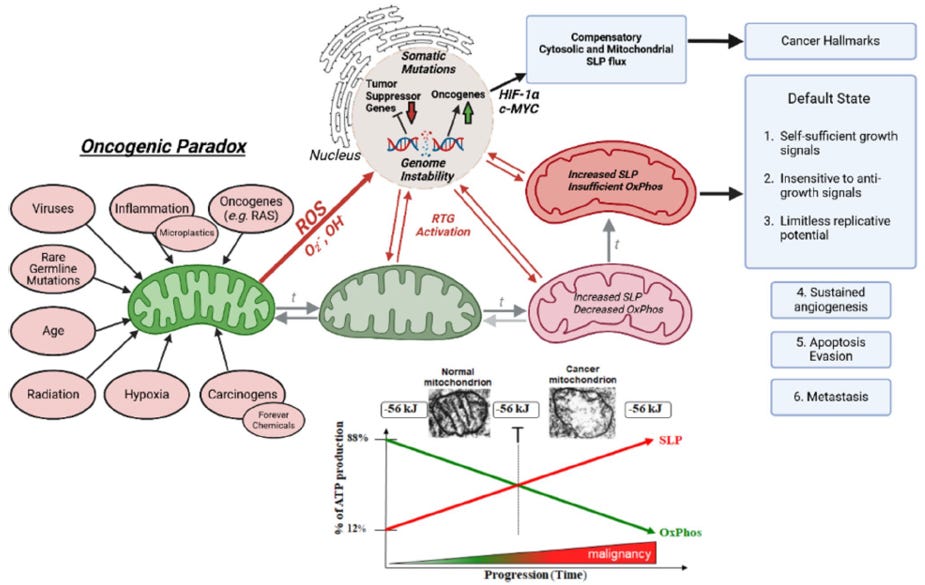
Although very specific processes underlie malignant transformation, many non-specific influences can initiate diseases — including radiation, chemicals, viruses, inflammation, etc. Indeed, it appears that prolonged exposure to almost any provocative agent in the environment can potentially cause cancer. (10) That a very specific process could be initiated in very unspecific ways was considered “the oncogenic paradox” by Szent-Gyorgyi. (10) This paradox has until recently been unresolved. (11) Abnormal mitochondrial structure and function is a universal finding in all cancer cells, as systematically documented by Dr Thomas Seyfried (see Figure 1).
Still, the concept of genetic mutations and genetic instability underpins most conventional cancer treatments. Big Pharma and the medical establishment have propagated this concept to promote the use of very expensive and toxic chemotherapeutic drugs; as mentioned above, cancer is profitable for the pharmaceutical industry. Curing cancer is not the goal.
There is considerable evidence that the genetic mutation theory may not be entirely correct. Dr. Thomas Seyfried provides a compelling argument that cancer is primarily a metabolic rather than a genetic disease. (11, 12) His underlying hypothesis is that cancer is a mitochondrial disorder with impaired oxidative phosphorylation and energy production; the genomic abnormalities are likely secondary to disordered energy production and cellular metabolism (bi-directal feedback; see Figure 1). Dr. Seyfried has clearly demonstrated that disordered mitochondrial function and energy production are common to all cancers. (11, 12) The view of cancer as primarily a metabolic disease will dramatically impact the approach to cancer management and prevention. However, it is clear that a very complex and bi-directional relationship exists between genetic instability and mitochondrial dysfunction.
Figure 1. The Bidirectional mitochondrial-nuclear relationship in carcinogenesis.
Reproduced from Seyfried TN, et al. J Bioenerg Biomembr 2025; 57:57-83
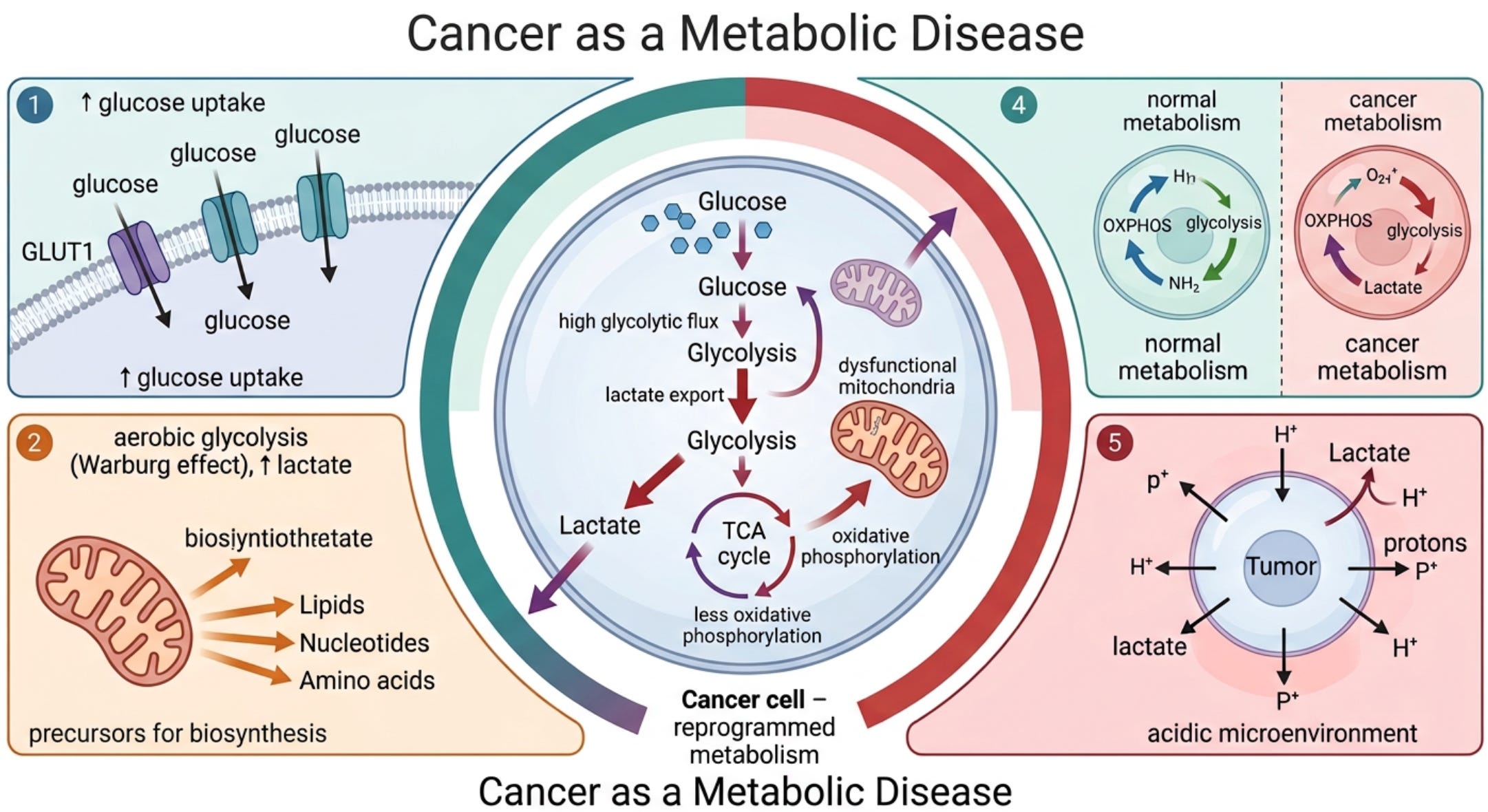
The idea that cancer is a metabolic disease was first noted by Otto Warburg in 1927, who was awarded the Nobel Prize in Medicine and Physiology in 1931 for his discoveries. (3, 4) Dr. Warburg, reported that cancer cells are dependent on aerobic glycolysis (breakdown of glucose to lactate) with impaired oxidative phosphorylation (pyruvate does not enter the Krebs cycle in the mitochondria). (3, 4) In simple terms, this means cancer feeds on glucose (see figure 2).
In contrast to normal differentiated cells, which rely primarily on mitochondrial oxidative phosphorylation to generate the energy needed for cellular processes, most cancer cells instead rely on aerobic glycolysis, a phenomenon termed “the Warburg effect.” (13) Dr. Warburg proposed that irreversible damage to respiration was the prime cause of cancer. Aerobic glycolysis in cancer cells involves elevated glucose uptake with lactic acid production in the presence of oxygen. (11)
Following his extensive research on tumor metabolism, Dr. Warburg stated: “Cancer, above all other diseases, has countless secondary causes. But, even for cancer, there is one prime cause. Summarized in a few words, the prime cause of cancer is the replacement of the respiration of oxygen in the normal body cell by fermentation of sugar.” (3, 4)
This metabolic phenotype is the basis for tumor imaging using labeled glucose analogs and has become an important diagnostic tool for cancer detection and management. Genes for glycolysis are overexpressed in the majority of cancers examined. (11) Numerous studies show that tumor mitochondria are structurally and functionally abnormal and incapable of generating normal levels of energy. (14-19) In addition, there is compelling evidence that mitochondrial dysfunction, operating largely through the RTG response (mitochondrial stress signaling), underlies the mutator phenotype of tumor cells (see figure 1). (20-24) Impaired mitochondrial function can induce abnormalities in tumor suppressor genes and oncogenes.
Figure 2. The Warburg effect
The Warburg effect is characterized by metabolic reprogramming in cancer cells, favoring glycolysis over oxidative phosphorylation even under aerobic conditions. This shift involves upregulation of glycolytic enzymes and downregulation of tumor suppressors that normally restrict glycolysis
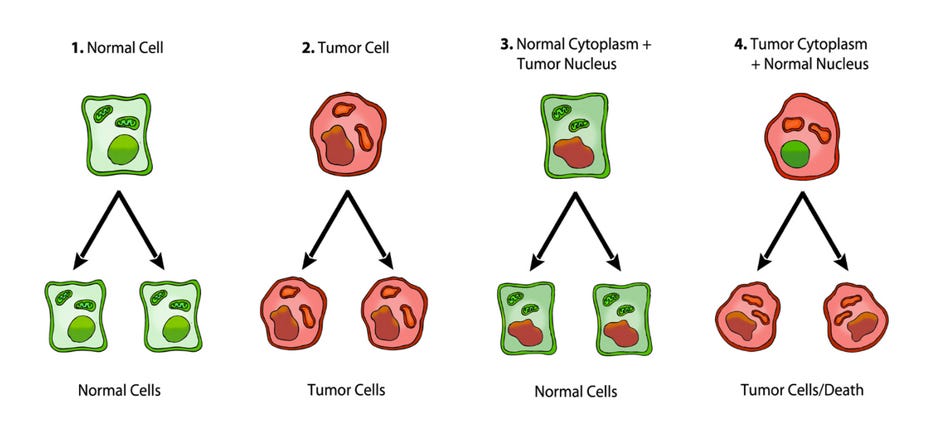
It is well documented that tumorigenicity can be suppressed when cytoplasm from enucleated normal cells is fused with tumor cells to form cybrids, suggesting that normal mitochondria can suppress the tumorigenic phenotype. (25, 26) Singh and co-workers provided additional evidence for the role of mitochondria in the suppression of tumorigenicity by showing that exogenous transfer of wild-type mitochondria to cells with depleted mitochondria (rho0 cells) could reverse the altered expression of the APE1 multifunctional protein and the tumorigenic phenotype. (27) It is also well documented that nuclei from cancer cells can be reprogrammed to form normal tissues when transplanted into normal cytoplasm, despite the continued presence of the tumor-associated genomic defects in the cells of the derived tissues (figure 3). (28, 29)
Figure 3. Nuclear and mitochondrial transplant experiments
Viruses have long been recognized as the cause of some cancers. It is interesting that several cancer-associated viruses localize to, or accumulate in, the mitochondria (including SARS-CoV-2). Viral alteration of mitochondrial function could potentially disrupt energy metabolism, thus altering expression of tumor suppressor genes and oncogenes over time. Viruses that can affect mitochondrial function include the Epstein-Barr virus (EBV), Kaposi’s sarcoma-associated herpes virus (KSHV), human papillomavirus (HPV), hepatitis B virus (HBV), hepatitis C virus (HCV), and human T-cell leukemia virus type 1 (HTLV-1) as well as SARS-CoV-2. (30-32)
A cell’s first line of defense against becoming cancerous is apoptosis. The apoptotic pathway is kept in check by anti-apoptotic factors; these two systems function in balance, and when one or the other becomes dominant, the cell either apoptoses, or it resists apoptotic signals. The metabolic approach to cancer treatment promotes apoptotic pathways.
In addition to the ultrastructural abnormalities in mitochondria and mitochondrial-associated membranes, no cancer cell has been found with a normal content or composition of cardiolipin, the cristae-enriched phospholipid that contributes to oxidative phosphorylation (OxPhos). Cardiolipin is recognized as essential for the proper function of the electron transport chain (ETC) super complex structures, which are linked directly to cristae ultrastructure. (33) Apart from these documented abnormalities in mitochondria structure and function, genetic abnormalities that alter mitochondrial function have also been recognized in many cancers. The p53 mutation, which is found in many cancers, can disrupt mitochondrial OxPhos. The retinoblastoma tumor suppressor protein, Rb, has been linked to abnormalities in mitochondrial mass and OxPhos function. (33) It appears that few, if any, cancer types are free of mitochondrial abnormalities, whether structural or functional, making OxPhos inefficiency the signature metabolic hallmark of cancer. As tumor cells require a significant ATP/ADP ratio for invasion, an alternative system for ATP synthesis must be in place to compensate for OxPhos inefficiency. A reliance on cytoplasmic (glycolysis) and mitochondrial substrate level phosphorylation (SLP) can provide both the necessary ATP and the metabolic building blocks needed for tumor cell proliferation and invasion in either aerobic or anaerobic growth environments. (33)
Cells using oxygen consumption for ATP synthesis will die quickly under hypoxia or when treated with cyanide. As many cancer cells can survive when treated with cyanide or in hypoxia, ATP synthesis in these cells must come from sources other than OxPhos. (33) The genomic instability and random somatic mutations seen in most cancers arise largely as downstream epiphenomenon of ROS production and OxPhos dysfunction.
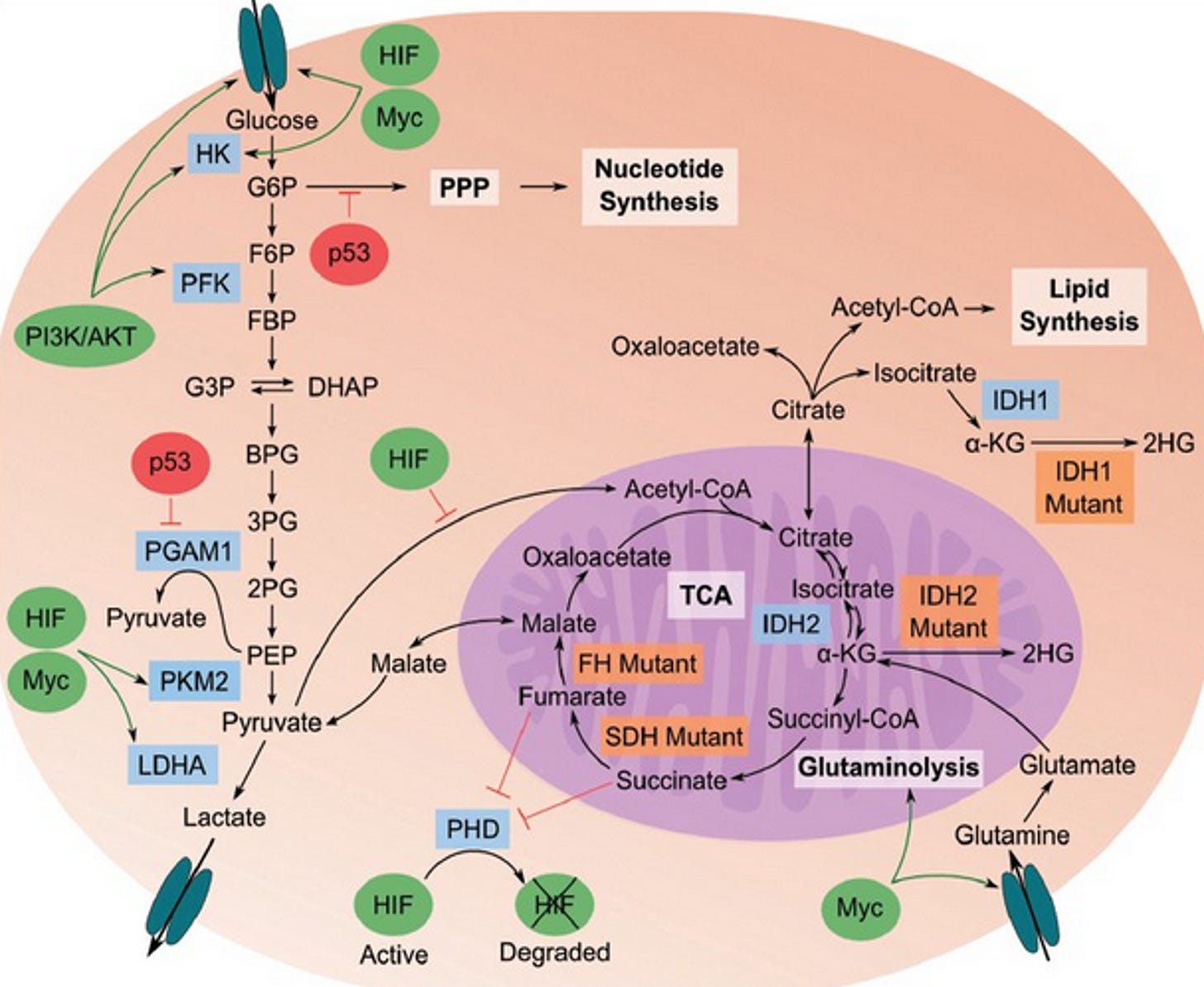
Since the 1950s, it has been recognized that tumors require large amounts of glutamine for growth and survival (hence the inclusion of glutamine in most culture media). The high-affinity glutamine transporter Slc1a5 (ASCT2) is upregulated in multiple types of cancer including glioblastoma multiforme (GBM) and has been implicated in mediating net glutamine uptake. (34) Several decades later, it was recognized that glutamine is a major energy source in tumor cells including GBM. (11, 12, 33-36) The interconversion of glutamine and glutamate is bidirectional in normal cells, with glutamine synthetase catalyzing glutamine formation. In tumors, however, overexpression of glutaminases and suppression of glutamine synthetase favor the forward reaction toward glutamate. Glutaminase activity correlates well with tumor growth rates in vivo. Glutamine not only provides nitrogen for synthesis of nucleotides and NEAAs but also provides a-ketoglutarate to serve as a precursor for ATP synthesis through substrate-level phosphorylation in the citric acid cycle.
Abnormalities in the cancer cell mitochondrial network would reduce OxPhos efficiency, thus forcing the cell to rely more heavily on SLP for ATP synthesis. The succinate-CoA ligase (SUCL) is a mitochondrial matrix enzyme that catalyzes the conversion of succinyl-CoA and ADP to CoA-SH, succinate, and ATP. Notably, when the SUCL proceeds toward ATP formation it is termed ‘‘mitochondrial substrate-level phosphorylation” (mSLP), a process that can yield high-energy phosphates in the absence of oxygen. Energy generation through mSLP is critically important in several metabolic pathways and could compensate for inefficient energy production through Ox-Phos in cancer cells. The glutaminolysis pathway would support production of high-energy phosphates through the sequential metabolism of glutamine → glutamate → α-ketoglutarate → succinyl CoA → succinate. (11, 12, 33-36) Glutamine has long been considered an essential metabolite for tumor cell growth. (37) Glutaminase is an enzyme that catalyzes the production of glutamate from the amino acid glutamine, which then feeds into the TCA cycle.
Chen et al. showed that glutamine utilization is a common feature of cells with partial defects in OxPhos, irrespective of the specific OxPhos complex affected. (38) OxPhos inefficiency could account in large part for the glutamine addiction of cancers. Glutamine-supported mSLP can compensate for OxPhos deficiency in either hypoxic or normoxic growth environments. It is well recognized that most, if not all, tumor cells are dependent on glucose and glutamine for growth. Although amino acids other than glutamine can also provide energy through mSLP, glutamine is the only amino acid not requiring expenditure of energy for the metabolic interconversions necessary to produce succinyl-CoA. (33)
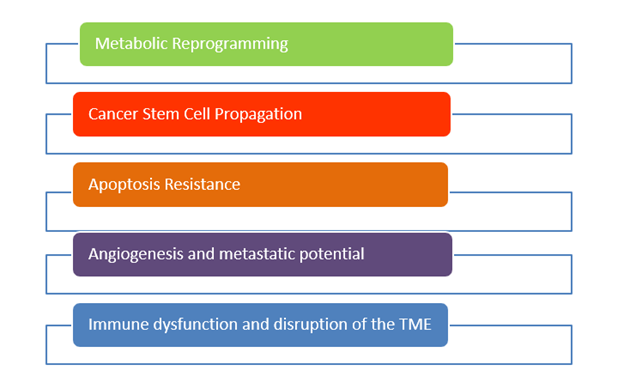
Mitochondrial substrate level phosphorylation (mSLP) in the glutamine-driven glutaminolysis pathway, substantiated by the succinate-CoA ligase reaction in the TCA cycle, can partially compensate for reduced ATP synthesis through both Ox- Phos and glycolysis. A protracted insufficiency of OxPhos coupled with elevated glycolysis and an auxiliary, fully operational mSLP, would cause a cell to enter its default state of unbridled proliferation with consequent dedifferentiation and apoptotic resistance, i.e., cancer. (33) The simultaneous restriction of glucose and glutamine offers a therapeutic strategy for managing cancer. The revised hallmarks of cancer as formulated by Marik & Hope is illustrated in Figure 4.(39)
Insulin and cancer
Insulin, insulin-growth factor-1 (IGF-1), phosphoinositide 3-kinase (PI3K) and mTOR are nutrient sensors and cellular growth factor associated with initiation and propagation of cancer.(40) Established risk factors for cancers include obesity, sedentary lifestyle and type 2 diabetes mellitus which are characterized by hyperinsulinemia and insulin resistance. (41) Higher circulating insulin and C-peptide (a marker of insulin resistance and long-term insulin secretion) have also been associated with an increased risk of cancer. The association between hyperinsulinemia and cancer suggests that a diet inducing an elevated insulin response may contribute to tumour growth. A recent study showed that higher dietary glycemic load was associated with an increased risk of recurrence and death in stage III colon cancer patients. (42) Yuan et al determined the association of post-diagnosis dietary insulin scores with survival among 2006 patients from two large prospective cohorts who were diagnosed with colorectal cancer.(41) The insulin score was developed to quantify postprandial insulin response for various food items. In the study by Yuan et al the adjusted HRs for colorectal cancer specific mortality comparing the highest to the lowest quintiles of the dietary insulin load was 1.82 (95% CI: 1.20–2.75, p=0.006).
Figure 4. Revised 2026 Hallmarks of Cancer (Marik & Hope).(39)
CANCER SIGNAL PATHWAYS
Signaling pathways are a core system in which cells regulate various physiological processes and respond to external stimuli. Normally, cells have a complete set of regulatory mechanisms for initiating and/or inhibiting signal reception, cascade transmission, and ultimately gene expression, but in cancer cells, the signaling pathway is usually overactivated, and the balance is broken. Almost all the nutraceutical and repurposed drugs listed which we recommend have anticarcinogenic effects by promoting and/or inhibiting signal transmission through the targeted regulation of multiple links in important signal pathways. The most relevant pathways include the following:
Hexokinase-2 (HK2) pathway. In 1977, Pete Pedersen isolated the metabolic defect responsible for the Warburg effect: the hijacking of normal hexokinase by hexokinase II (HK2), followed by its massive overproduction. (8, 43) Hexokinase is the first step in glycolysis in the cytoplasm. Cancer cells have switched to an embryonic form of hexokinase (HK2), which then translocates from the cytoplasm to the outer mitochondrial membrane, where it is attached to the voltage-dependent anion channel (VDAC). (44-46) The VDAC is a pore-like opening in the outer membrane involved in shuttling nutrients and signaling molecules in and out of the mitochondria. HK2 is the major bound hexokinase isoform expressed in cancer cells that exhibit the Warburg effect. By stationing itself on the outer mitochondrial membrane, HK2 helps immortalize cancer cells, escapes product inhibition, and gains preferential access to newly synthesized ATP for phosphorylating glucose. (47) The attachment of HK2 to the VDAC on the outer mitochondrial membrane creates a state of apoptosis resistance and shunts ATP out of the mitochondria to the cytoplasm to support glycolysis. When bound to HK2, the VDAC gate is “locked”, preventing the release of cytochrome c, thereby preventing apoptosis, and effectively immortalizing the cell. Several drugs target HK2, separating the enzyme from the outer mitochondrial membrane; these include 3-bromopyruvate, curcumin, resveratrol, and its derivatives pterostilbene and quercetin.
The p53 pathway (the tumor suppressor pathway). (48) The p53 pathway is activated by sensor kinases which monitor the cell’s DNA for damage or errors. Upon detection of damage, they phosphorylate the nuclear localization factor of the p53 tumor suppressor protein, allowing it to translocate to the cell nucleus and begin expressing p21, p16, p15, and p19; this activates the cell cycle arrest pathway, initiating DNA repair, and preventing cell division. If the repair is deemed to have failed, BAX, BAK, and/or PUMA are expressed, among others, initiating the mitochondrial caspase cascade, which initiates apoptosis.
The TGF-β pathway. The TGF-β pathway plays a crucial role in regulating cell growth, differentiation, and apoptosis. (49) Upon binding to its cell surface receptors, TGF-β activates SMAD transcription factors, leading to the repression of anti-apoptotic genes and the activation of pro-apoptotic genes. This pathway acts as a tumor suppressor by promoting apoptosis in abnormal cells and inhibiting the growth of precancerous cells. Defects in this pathway can lead to uncontrolled cell growth and the development of cancer.
The Wnt signaling pathway. The Wnt/beta-catenin pathway is a family of proteins that is implicated in many vital cellular functions such as stem cell regeneration and organogenesis.
The Wnt signaling pathway plays a crucial role in the regulation of cell proliferation and differentiation. (50) In normal conditions, Wnt signaling maintains the balance between cell proliferation and apoptosis to ensure healthy tissue growth. However, when the pathway is activated excessively or inappropriately, it can lead to the development of cancer. Wnt activation has been observed in breast, lung, and hematopoietic malignancies and contributes to tumor recurrence.(51) The Wnt pathway cross talks with the Notch and Sonic Hedgehog pathways, which has implications for therapeutic interventions in cancers.
The Notch signaling pathway. The Notch signaling pathway is a signaling mechanism that plays a role in cell differentiation, proliferation, and apoptosis. (52) Disruptions in the Notch pathway, such as mutations in Notch receptors or ligands, can lead to the dysregulation of cell proliferation and differentiation, contributing to the development of cancer. Notch signaling plays a crucial role in the development of colon cancer; targeting the Notch pathway may sensitize colon cancers to various adjuvant agents.(53)
The PI3K/AKT signaling pathway. The phosphoinositide 3-kinase (PI3K) signaling pathway is linked to both growth control and glucose metabolism. The activation of the PI3K/AKT signaling pathway occurs when growth factor receptors on the cell surface bind to their ligands, triggering the activation of PI3K. (54) Once activated, AKT phosphorylates and inhibits the activity of pro-apoptotic proteins, such as BAD and FOXO. AKT also activates mTORC1, which regulates cellular metabolism and promotes cell survival by stimulating the expression of anti-apoptotic genes Bcl-2 and Bcl-xL.
The Hedgehog Pathway. Hedgehog (Hh) is one of the few signaling pathways that is frequently used during development for intercellular communication. (55) Hh is important for the organogenesis of almost all organs in mammals, as well as in regeneration and homeostasis. Further, Hh signaling is disrupted in diverse types of cancer. Mebendazole decreases the activity of the Hedgehog pathway, which is common in gliomas, melanomas, lung cancers, ovarian cancers, and colorectal cancer. (56)
The insulin growth factor-1 (IGF-1) pathway. Insulin-like growth factor 1 (IGF-1) is produced primarily by the liver as an endocrine hormone, as well as in target tissues in a paracrine/autocrine manner. IGF-1 signaling is mainly mediated by binding to its specific receptor, the insulin-like growth factor 1 receptor (IGF-1R) leading to the activation of the AKT signaling pathway resulting in cell growth, proliferation, and inhibition of programmed cell death. An elevated level of circulating IGF-1 is an established risk factor for many cancer types, whereas a decrease in IGF-1 levels is associated with lower cancer incidence.
Now that you have a basic understanding of cancer biology the next post will build upon this knowledge and explore the metabolic approach to treating cancer.
This Substack had previously written about the “Warburg effect,” which is why diet is so vital when treating cancer since these metastasizing deadly cells depend on anaerobic glycolysis, even when oxygen is present; for example:
In fact, the following treatment approach may not only be the ‘holy grail’ cancer cure in plain sight, but may also treat Alzheimer’s, mood disorders, Parkinson’s, Lyme Disease, myocarditis, Hashimoto’s Disease, leukemia, Lupus, skin conditions, and various other “incurable” ailments, as well as the common cold and seasonal flu:
The Ultimate Disease Cure & Prophylaxis Protocol
Tocotrienol and Tocopherol forms (all 8) of Vitamin E (400-800mg per day, 7 days a week). A product called Gamma E by Life Extension or Perfect E are both great.
Bio-Available Curcumin (600mg per day, 2 pills per day 7 days a week). A product called Theracurmin HP by Integrative Therapeutics is bioavailable.
Vitamin D (62.5 mcg [2500 IU] seven days a week).
CBD oil (1-2 droppers full [equal to 167 to 334 mg per day] under the tongue, 7 days a week) CBD-X: The most potent full spectrum organic CBD oil, with 5,000 milligrams of activated cannabinoids and hemp compounds CBD, CBN & CBG per serving.
Fenbendazole (300mg, 7 days a week) or in the case of severe turbo cancers up to 1 gram — for prophylaxis one 150mg tablet once or twice per week
Ivermectin (24mg, 7 days a week) or in the case of severe turbo cancers up to 1mg/kg/day — for prophylaxis one 12mg tablet once or twice per week
Hydroxychloroquine (10mg/kg/day 7 days a week) - for prophylaxis one 200mg tablet once or twice per week
Doxycycline (100mg, 7 days a week for 30-60 days)
VIR-X immune support which also greatly increases the bioavailability of both Fenbendazole and Hydroxychloroquine (2 capsules per day) — for prophylaxis 2 capsules per day
Removing sugars and carbohydrates (cancer food) from your diet and replacing table sugar with a zero glycemic index, zero calorie, keto friendly rare sugar like FLAV-X
And here is additional information on why Hydroxychloroquine was also added to this protocol as a most viable anticancer compound:
The Medical-Industrial Complex does NOT want you to know about this disease cure-all protocol.
Do NOT comply.
‼️ THE 25% OFF LIFESAVING FLASH SALE ends in a matter of hours, so please use code IVM25 to receive 25% off on the newest product Hydroxychloroquine, as well as ALL of the amazing lifesaving products that you have been purchasing for many years now like the Nobel Prize winning miracle compound Ivermectin, the no less miraculous Fenbendazole, Doxycycline, the full spectrum organic CBD oil containing 5,000 milligrams of activated cannabinoids and hemp compounds CBD, CBN & CBG, the powerful immune support nutraceutical and spike support formula VIR-X, and the sugar craving reducing, blood sugar balancing and even anti-cancer allulose sugar substitute FLAV-X! ‼️
THE MASSIVE 25% OFF WEEKEND FLASH SALE ends TONIGHT, March 8th (midnight eastern time), 2026.
Upon adding products to your cart, please go to the cart icon at the top right corner of your browser page and click it, then choose the VIEW CART option whereby you will be redirected to a page where you can enter the code IVM25 in the Use Coupon Code field.
Only the RESOLVX HEALTH website offers all of the authentic products that this Substack promotes.
Please contact the company directly with any product questions: info@resolvx.health







A number of doctors/researchers have reached this conclusion and built therapies on that basis. See for example German Researcher Otto Warburg's early work on the theory that all cancer is a disease of energy metabolism; the many fine books, and metabolic therapies of Dr. Nicholas Gonzalez with a central element of cancer-countering pancreatic enzymes; layman Chris Wark and his diet and exercise emphasizing program Chris beat cancer; Winters et al, in their book, "The Metabolic Approach to Cancer," and the modern research base established in Thomas Seyfried's book Cancer as a Metabolic Disease, which draws heavily on Warburg's work.
The key issues are a) getting the word out while battling the propaganda of the powers that be, and especially, b) helping people actually believe it, given that they are "pre-programmed" or hypnotized by the medical establishment to believe that cancer is genetic and can only be addressed with cut, burn, and poison. And of course people have to be active in re-balancing their bodies, not passive by-standers.
Dr thank you for sharing this most wonderful stack today. Could emotions, toxins, spirit have effect how the cells become inefficient yet don't die off as normal would? Could glycine and glysophate misfolded be contributing factor. Glysophate antibiotic and microbiome immune function play a roll in factor. How critical is the energy of the electron transport glutathione and cholesterol. Cells adapt to their environment.
Fasting H2O, red light all frequencies, grounding or earth bathing reduce 5g etc. I remember Hilda Clark most cancer misdiagnosed are parasite infection.
The most shocking was the brain choose sugar over omega 3 good fats could this be culprit for auto immune disorders dementia Parkinson's ms especially processed high fructose from genetically modified or bioengineering.
If they say to give pet or live stock treatment every month and humans don't. Please test what is in the gut if you need antibiotics or additional support from botanicals bitters teas tinctures herbs. Please make sure your pathways of elimination are open. Get whatever minerals vitamins you are missing low and slow also take the time away from. Rest and digest so body can clean up brain body. If in fight flight freeze makes detox of body longer. Good food fermented food a small teaspoon approximately or smaller diversity of gut. Good thoughts gratitude prayer meditation
Filter air and possible plants to help filter. Filter water ewg had more information. Farmers markets or even grow food in water at home. There are resources there to help one in their journey in life no matter what. You are a great human and no one knows when you called. So if a human say you expire this or that don't believe it research your answers and choose what you wish to do.
There are rife frequencies, light, lazer, pemf, grounding mats, tibetan singing bowls, any many other things to help humans even pets too to live happy healthy lives.
You are the captain of your vessel your free to plan a course and go do it. If you need guidance ask medical people, elders, holy people. It's great today many disciplines are combined for a better outcome for you.
Today it's been highly recommended that you take care of the mouth 3D cone beam scan more sensitive to find hidden infections especially in root canal done. There has been some connection between oral microbiome and infection, disease, and cancer.