DEMENTIA & ALZHEIMER'S CURE: Fungal Infection in the Brain Produces Effects Similar to Alzheimer's
Some researchers have suggested that neurodegenerative diseases like Alzheimer's, Parkinson's, and multiple sclerosis may have a microbial origin; thus, inexpensive repurposed drugs may be the cure.
A recent article by The Epoch Times posited that fungal infections may be the cause of neurodegenerative diseases.
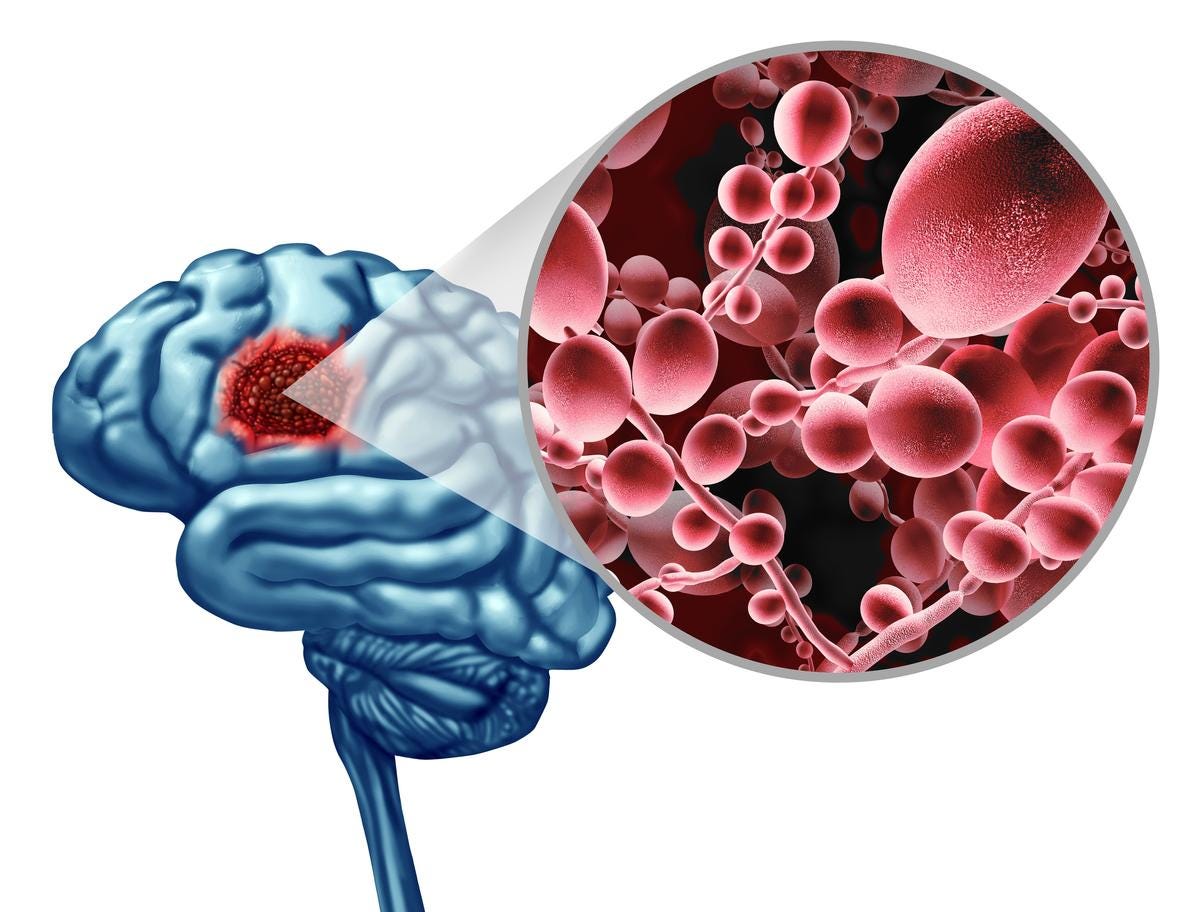
A team of researchers at Baylor College of Medicine has discovered that when the brain is infected with a common fungus, it changes in ways similar to those seen in Alzheimer’s disease. The new research delves deeper into some of the molecular mechanisms behind that process.
The Study Findings
Using animal models, the research team discovered how the fungus, called Candida albicans (C. albicans), enters the brain, activates mechanisms for its clearance, and generates amyloid beta (Aβ)-like peptides—toxic protein fragments thought to be central to the development of Alzheimer’s disease.
The findings were published Oct. 10 in the journal Cell Reports.
Previous research has implicated fungi in the development of chronic neurodegenerative diseases like Alzheimer’s disease, but their mechanisms are not entirely understood.
We know that Candida thrives on sugars, so diets high in processed foods and junk foods may very well be feeding the funguses that in turn destroy brains.
We also know that the inexpensive and repurposed drug Fenbendazole (Mebendazole being the analog drug with the addition of a single molecule which adds nothing save for bogus patent-based profits) eradicates these funguses; for example, a research study entitled, The Anti-helminthic Compound Mebendazole Has Multiple Antifungal Effects against Cryptococcus neoformans stated the following:
Cryptococcus neoformans is the most lethal pathogen of the central nervous system. The gold standard treatment of cryptococcosis, a combination of amphotericin B with 5-fluorocytosine, involves broad toxicity, high costs, low efficacy, and limited worldwide availability. Although the need for new antifungals is clear, drug research and development (R&D) is costly and time-consuming. Thus, drug repurposing is an alternative to R&D and to the currently available tools for treating fungal diseases. Here we screened a collection of compounds approved for use in humans seeking for those with anti-cryptococcal activity. We found that benzimidazoles consist of a broad class of chemicals inhibiting C. neoformans growth. Mebendazole and fenbendazole were the most efficient antifungals showing in vitro fungicidal activity. Since previous studies showed that mebendazole reaches the brain in biologically active concentrations, this compound was selected for further studies. Mebendazole showed antifungal activity against phagocytized C. neoformans, affected cryptococcal biofilms profoundly and caused marked morphological alterations in C. neoformans, including reduction of capsular dimensions. Amphotericin B and mebendazole had additive anti-cryptococcal effects. Mebendazole was also active against the C. neoformans sibling species, C. gattii. To further characterize the effects of the drug a random C. gattii mutant library was screened and indicated that the antifungal activity of mebendazole requires previously unknown cryptococcal targets. Our results indicate that mebendazole is as a promising prototype for the future development of anti-cryptococcal drugs.
Thus, we may extrapolate that Fenbendazole, being a powerful antifungal, may quickly and permanently reverse these kinds of neurodegenerative diseases, especially since, “…mebendazole reaches the brain in biologically active concentrations…”
And precisely because C. albicans has been shown to generate toxic protein fragments like amyloid beta (Aβ)-like peptides in the brain, Fenbendazole would be an especially strong candidate for potential therapeutic use in Alzheimer's treatment.
Back to The Epoch Times article, which reinforces this notion of fungal-induced cognitive decline:
Humans and C. albicans have a complicated relationship, as most of the time, C. albicans is harmless and is simply a member of a healthy microbiota. However, it is one of the few fungal species that cause disease in humans and is responsible for infections that range from superficial infections of the mucosa and skin, like thrush, diaper rash, and vaginal yeast infections, to more serious infections like invasive candidiasis that can affect the blood, heart, brain, and bones.
Infections by C. albicans are particularly dangerous for those with compromised immune systems, like those with AIDS, or people undergoing immunosuppressive therapies for cancer and other conditions. This suppression of the body’s defenses is part of the reason why some people acquire C. albicans infections after taking antibiotics, as they decrease the beneficial bacteria in the gut, causing an imbalance and allowing C. albicans to thrive. C. Albicans can survive outside the body and have the ability to colonize every human organ and tissue. According to the Encyclopedia of Microbiology, it is the most common cause of systemic fungal infections.
Since so much of humanity is now suffering from (subclinical) VAIDS, infections by C. albicans will become far more prevalent, as will the associated cases of Modified mRNA “vaccine”-induced early onset dementia and Alzheimer's.
In a January review published in Frontiers in Immunology, researchers examined the role of fungus in central nervous system autoimmune and neurodegenerative disorders. The review states that recently, increasing evidence has pointed to the role of peripheral fungus in triggering inflammation, immune response, and worsening of a range of non-infectious disorders of the central nervous system (CNS), including multiple sclerosis, Parkinson’s, and Alzheimer’s disease.
The review concluded that fungus can trigger inflammation via different mechanisms in the progression of CNS non-infectious diseases, suggesting that it is crucial for developing future therapeutic agents and strategies.
A 2015 study published in Scientific Reports states that several researchers have proposed the possibility that Alzheimer’s disease may have a microbial cause. The researchers found evidence that tissue from the central nervous system (which includes the brain and spinal cord) of patients with Alzheimer’s disease contains fungal cells. These fungal cells were found in different regions of the brain, including the external frontal cortex, cerebellar hemisphere, entorhinal cortex/hippocampus, and choroid plexus. These fungal materials were not present in the control individuals who did not have Alzheimer’s disease. The researchers identified several different species of fungus in their samples.
What is of particular note are the surges of both blood clots and immunocompromisation coinciding with the DEATHVAX™ rollout, and the consequent increases of fungal infection susceptibility contributing to ever more deadly outcomes:
Interestingly, the study authors noted that fungal infection was also found in the blood vessels, which could explain the vascular pathology that is often found in Alzheimer’s patients.
Once again, the confluence of “vaccine” adverse events is truly curious.
The findings provide intriguing evidence that these fungal infections are present in the central nervous systems of those with Alzheimer’s disease and not in healthy individuals who served as the controls.
Given that over time the slow kill bioweapon injections render increasingly larger swaths of unhealthy individuals, the unvaccinated healthy individuals will serve as the ultimate controls, especially for increasingly prevalent diseases such as dementia and Alzheimer’s.
Another Piece of the Puzzle
Dr. Corry and his colleagues have provided another piece to further understand the role fungus may play in the development of Alzheimer’s disease.
“This work potentially contributes an important new piece of the puzzle regarding the development of Alzheimer’s disease,” Corry said in a press release. “The current explanation for this condition is that it is mostly the result of the accumulation of toxic Aβ-like peptides in the brain that leads to neurodegeneration. The dominant thinking is that these peptides are produced endogenously [internally], our own brain proteases break down the amyloid precursor proteins generating the toxic Ab peptides.”
Due to their anti-prion activity, the other pieces of the therapeutic puzzle are Ivermectin and Quercetin.
Ivermectin, another antiparasitic drug, was shown by Kawasaki et al. (2007) to have prevented the formation of the infectious form of the prion protein in prion-infected cells. Meanwhile, Quercetin, a flavonoid, extended the lifespan of prion-infected mice, highlighting its therapeutic potential against prion diseases.
Both compounds have also exhibited properties that modulate tauopathy, a characteristic of Alzheimer's disease. Ivermectin inhibited tau hyperphosphorylation and even disaggregated tau fibrils. Quercetin, too, displayed capabilities against tauopathy by inhibiting the hyperphosphorylation of the tau protein.
The discovery of Fenbendazole, Ivermectin, and Quercetin's potential therapeutic properties underscores the importance of repurposing existing drugs in order to create highly accessible new treatment paradigms for neurodegenerative diseases.
Do NOT comply.






If anyone has links to specific protocols please post them. My aunt has Alzheimer's and has just been moved from my pharma uncle to a different, natural-minded aunt and we are trying to figure out what we can do to help her now. Thank you !
It's amazing how fast one's taste for sugar disappears when you stop eating sugar...and how clear the mind gets. Fermented and distilled sugars [read potato vodka] seem to be more tolerated. I would also like to point out the link between Nattokinase and Ivermectin. Both come from soil based elements, and both seem to have similar properties.